Let’s dive just a little deeper
Why Silicon ?
Silicon is the main material for the production of solar panels. It is the second most widespread element on the planet, and it can rarely be found in a free state in nature. It is most often found in the form of sand and minerals called silicates. Silicon belongs to the fourth group in the periodic system of elements, just like carbon. Carbon is the most important element in the organic world and silicon in the in organic. Similar to carbon, silicon has four electrons in its outer shell and forms four covalent bonds. Carbon is a good conductor of electricity. Silicon is a semiconductor, which makes it a good material for making PV cells. More about production process read here ….
Energy gap : Eg
All materials in nature can be conductors, semiconductors, or insulators depending on the number of electrons in the last shell and their bond energy. Silicon is a type of semiconductor. As the temperature rises, its electric resistance decreases. This is due to silicon’s small energy gap (band gap) between its highest occupied and lowest unoccupied energy levels (the valence band and the conduction band). The term “Fermi energy” refers to the energy difference between the highest and lowest occupied single-particle states in a quantum system of non-interacting fermions at absolute zero temperature. The Fermi level is located approximately halfway between the valence and conduction bands and represents the energy at which a state is equally likely to be occupied by an electron as not. As a result, pure silicon is an insulator at room temperature.
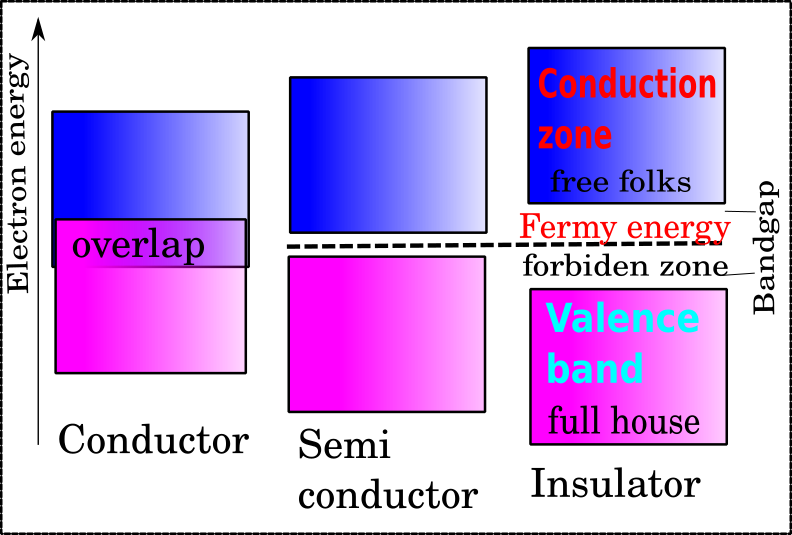
Simplified representation of Femi energy in conductors, semiconductors, and insulators
p-n mystery solved
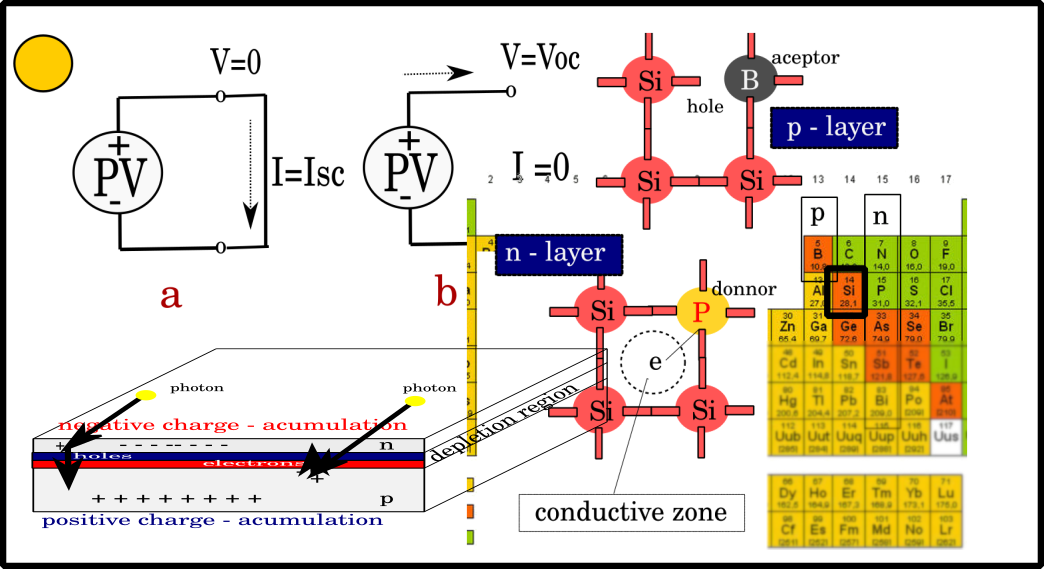
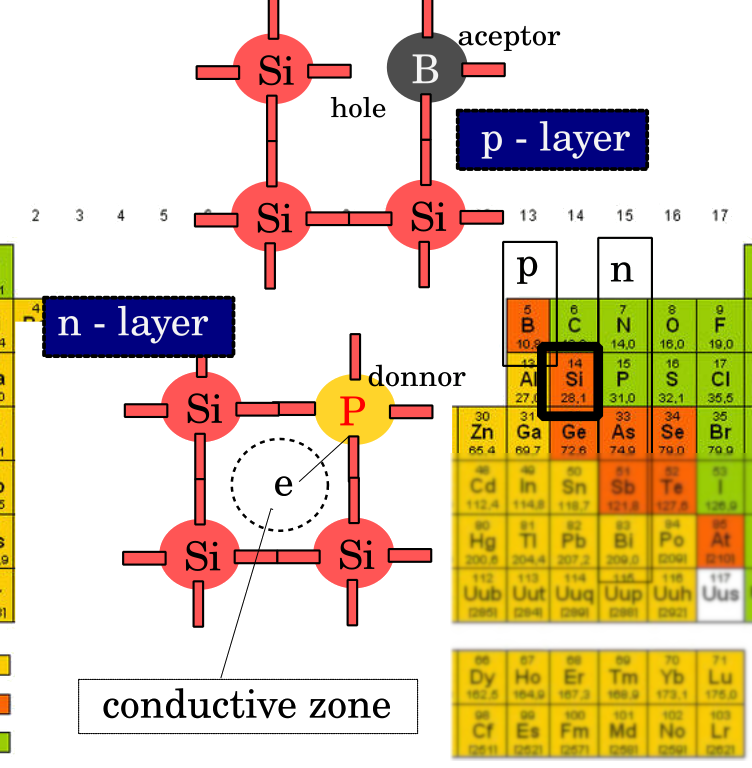
Elements of the 3rd group of the periodic table are used to form p-type semiconductors. By introducing boron into silicon, boron atoms replace individual silicon atoms in the crystal lattice. Since the boron atom is trivalent, it will form covalent bonds with three neighboring silicon atoms. The bond with the fourth neighboring silicon atom cannot be realized; the last electron from the back shell of silicon binds to the boron atom, which becomes negative, and the last silicon atom manifests the existence of a cavity, so that the total effect consists in the fact that excess voids are obtained in the doped material. Due to the fact that the boron atom accepted the last electron from the silicon shell, it is called an acceptor.
Elements of the 5th group of the periodic table are used to form an n-type semiconductor (for example, phosphorus). When a phosphorus atom replaces a silicon atom in the lattice, it will form covalent bonds with its neighbors, as with boron. The difference is that phosphorus, as a pentavalent element, after making bonds with four neighboring silicon atoms, will have one extra electron left. However, that last electron will not remain on the last shell of the phosphor but will be emitted into the conductive zone, i.e., one free electron is obtained. Due to the fact that pentavalent impurities cause an excess of negative charge to appear (which they “give”), they are called donors. The dopant material and their concentration in the semiconductor will depend on the desired characteristic of the requested p-n junction. Technically, a p-n junction is not made by connecting two different types of semiconductors by subsequent insertion of an admixture of the opposite type into the existing type.
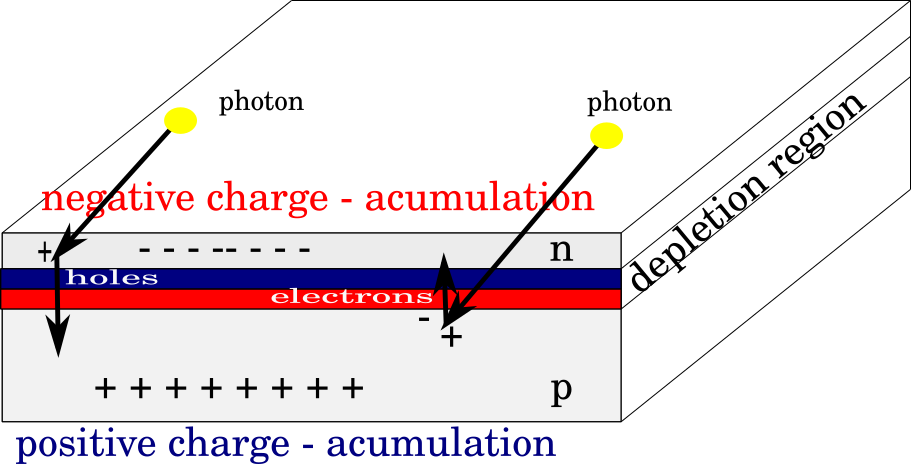
The semiconductor creates a p-n junction. Considering that it is a connection of two different types of semiconductors, one of which is redundantly negative, and that there is an excess of positive charges, there will be a diffusion movement of free electrons to the n-region and free cavities to the p-region. By passing free charges from one type into another, an electric field will be formed along the very border of the junction. That field will continue to represent an obstacle to the further diffusion of free carriers. The potential barrier thus formed is called the area of space charge. There will still be a tendency for free charges to move to the opposite side, but this will be prevented by the potential barrier, so the P-N junction is in dynamic balance.
Current is obtained
When the solar cell is illuminated, then photons will penetrate the material (N-type is a very thin layer) and collide with atoms near the space charge region. If the photon has a large enough energy to cause ionization of atoms, a new pair of electron-holes will be created under the influence 24 fields of the potential barrier to be transposed and the electron to n type, and the cavity to p type. In that manner, separation of charges occurs and voltage appears at the solar cell terminals. The basic condition for the creation of electron-hole pairs is that the energy of the photon that interacts with the crystal lattice in a semiconductor is sufficient to transfer electrons from the valence to the conduction zone, i.e., the following condition must be met:
hν≥Eg so we have : λ ≤ hc/Eg
where is:
λ – wave length
ν – frequency of incident photon
c – speed of light
h – Planck’s constant = 6.626×10-34 Js
Photons with wavelengths longer than the threshold λg=h*c/Eg are not absorbed in the solar cell. While absorbing photons with a shorter wavelength than the threshold, excess energy (h-Eg) passes into the crystal lattice, increasing her energy. The figure shows the spectrum of solar radiation (for AM = 1.5) and indicates part of the solar radiation energy that is theoretically converted into electrical energy for one silicon photo voltaic cell. From the above, it could be concluded that the wider the semiconductor, the more suitable it is for having a smaller energy gap because a semiconductor with a smaller Eg can absorb a larger range of wavelengths from the solar spectrum. However, the voltage of the solar cell also depends on EG. For higher EG, there is a smaller inverse saturation current through the p-n junction, i.e., a higher output voltage is obtained. So, semiconductors with a small energy gap are not suitable because the obtained voltages are too small. These two effects are opposite each other, so it is necessary to find an optimal solution. And that leads us to the next consideration:

The figure left shows the spectrum of solar radiation (for AM = 1.5) and indicates part of the energy of solar radiation that is theoretically converted into electrical energy for one silicon photovoltaic cell.
Figure right. Charges are separated and voltages appear as a result of photon action.
Open circle Voltage -Voc and Short circle current -Icc
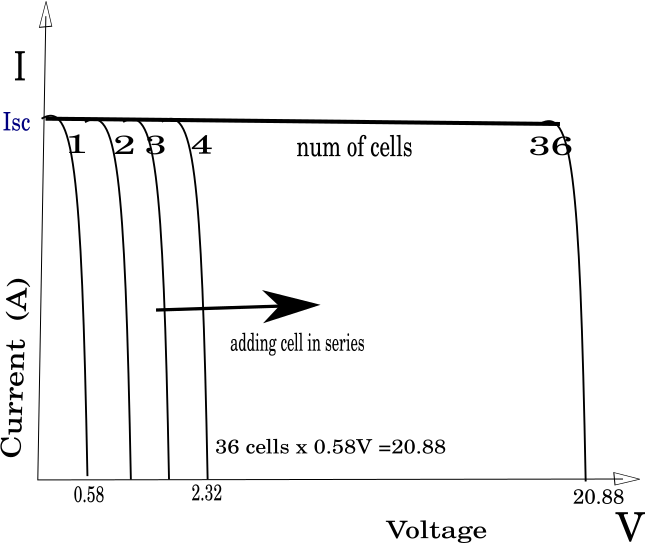
When the cell is illuminated, a current of charge carriers is generated through the p-n junction, similar to the previous one described, which is modeled by a current source on the photocell replacement scheme. Important parameters of a solar cell include open-circuit voltage (VOC) and short-circuit current (ISC) cells. The short-circuit current of the cell is obtained when the voltage at the output ends of the cell is equal to zero. Voc and Isc are functionally related. If the cell is illuminated with an irradiation of 1000 W/m2, the Isc will be about 4 A; with half the illumination of the cell, the Isc is reduced twice. The fruit will weaken slightly. The solar cells are connected to obtain the desired voltage and power. This is how solar is formed module. Several solar modules are connected suitable, and a solar panel is obtained.

a – Shortc circle current – no resistant found no voltage
b. Open circle: no current, maximum voltage
On the right: packing cells in series increases voltage (and power).
Conclusion
I tried to briefly and clearly explain the principle of operation of the solar cell. For more detailed information, it is necessary to consult the professional literature in that field.